 The second stage is the formation of the plastic material by polymerisation. (2)Ĭomplete the equation for the cracking of butane using displayed formulae.Ĭomplete the balanced chemical equation far the complete combustion of ethane in oxygen.C2.4(g) +. Butane (C4H10), a hydrocarbon in crude oil, can be cracked to produce two different hydrocarbons, ethane (C2.4) and ethene (C2H4) (i)įor cracking to happen what needs to be done to the hydrocarbon?. The first stage in the formation of a plastic material is called cracking. One reason the oil industry is important is that it uses crude oil to produce many of the plastic materials we use in everyday life. Other people say that increasing the production of ethanol from sugar cane will be bad for the environment. Some people say that increasing the production of ethanol from sugar cane will be good for the environment. The reaction to produce ethanol from sugar solution is Ethanol can be produced from sugar cane.ĭraw a ring around the correct answer to complete the sentence. Most petrol used in cars contains about 5% ethanol (C2H5OH). Complete the word equation for the complete combustion of octane. Petrol contains a mixture of compounds, including octane (C8H18). Methane and decane are gases at room temperature (20☌). Methane has the lowest melting point and icosane has the highest boiling point. ) two correct statements about the four compounds. The table below shows four of these compounds. This question is about compounds produced from crude oil. (i)Ĭomplete the equation below to show formation of the polymer from the monomer propene. 
Small molecules of substances called monomers can be joined together in polymerisation, eg. (2)Ī molecule of the compound methane, CH4, can be shown like this:ĭraw a molecule of the compound ethene, C2H4. 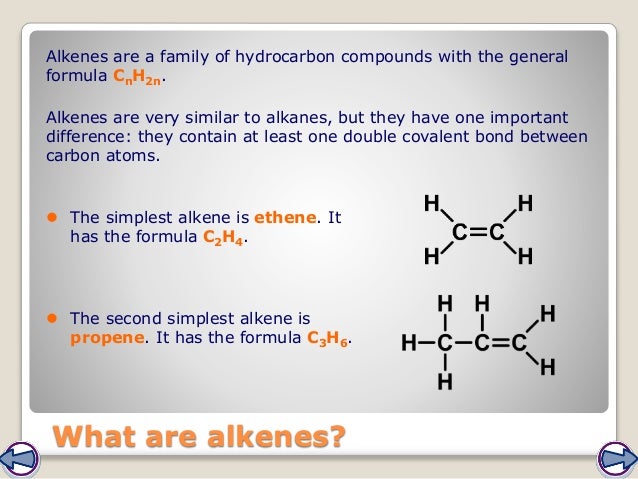
This type of reaction is carried out because there is a greater demand for the products than for the original hydrocarbon. 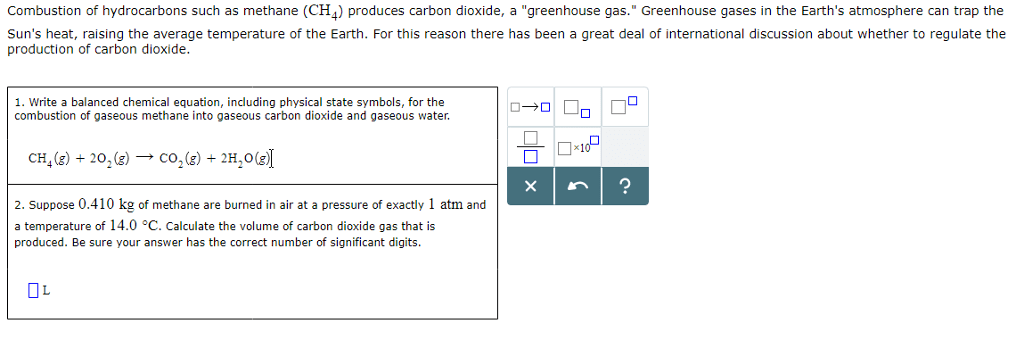
This is one of the reactions which took place: C16H34 The hydrocarbon C16H34 was heated strongly in the absence of air. What is the name of the process in which X is changed into poly(ethene)?. The hydrocarbon X is used to make poly(ethene). The high demand for petrol (octane) can be met by breaking down longer hydrocarbons, such as decane, by a process known as cracking.Īpart from heat, what is used to make the rate of this reaction faster?. Alkanes, alkenes, alcohols, cracking and polymers 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
